A Harder 22K Carat Gold Alloy

Abstract
After testing different alloy compositions, a 22-karat gold alloy was achieved that had both a similar color to that of the conventional yellow 22 karat gold alloy and a higher hardness. The developed alloy can be cast into intricate shapes, using common equipment and practice, and it can undergo in excess of 99% cold rolling without cracking or intermediate annealing. The hardness values of the new 22 karat alloy in the cast, cold rolled, and heat treated conditions are higher than those of conventional 22 karat gold, with a peak hardness of HV274 being achieved through a combination of cold rolling and heat treatment. The new alloy retains the excellent corrosion resistance of the conventional 22 karat alloy, and it has a similar tarnish behavior. No elements hazardous to human health are used in the alloy.
Background
The market price of gold has decreased over the last ten years while jewelry production has continued to grow, and different trends have emerged from the main jewelry producing areas of the world. In Europe, the most widely used alloys for jewelry are low to medium karat alloys such as 8, 9, 14, and 18 karat gold. In the United States, 14 karat gold predominates with some 10 and 18 kt, and in much of the Middle East, India and South East Asia, jewelers traditionally use 21, 22 and 24 karat gold. There is a large market for 22-karat alloys, especially in India and the Middle East, where 41% and 31% growth respectively, were recorded in 1997 (1). Other countries in Asia also use high-karat jewelry, which is seen as a way to save or invest money.
The main drawback of using high-karat gold jewelry is the softness of the material (2), which is more prone to wearing with time than, for example, an 18 karat alloy. A number of attempts have been made over the past few years to increase the hardness of high karat jewelry (3 – 5). Conventional 22 karat gold can only be hardened by cold working. Although can be cast, its hardness in the annealed condition is very low, HV52, (6).
The development of a harder 22-karat gold alloy is, therefore, a logical response to the problems posed by conventional 22 karat gold. Increasing its hardness would make it more wear and scratch resistant, more appropriate for machining and polishing, and probably more popular among other segments of the market.
Mintek has now developed such a 22-karat gold alloy. This alloy can be hardened to a value comparable with conventional 18 karat yellow gold. It contains 91.7 wt. % gold, and its color is very similar to that of conventional 22 karat yellow gold. The alloy was designed to meet the jewelers’ highest requirements and to comply with existing processing equipment and methods.
Hardness
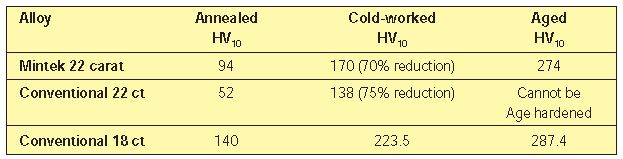
The conventional 22-karat gold alloy (91.7 wt. % Au – 5.5% Ag – 2.8% Cu) does not exceed a peak hardness of HV150 (6). Table 1 shows a comparison between Mintek’s alloy and conventional 18 and 22 karat gold alloys.
The peak hardness value achieved in Mintek’s alloy is the result of a combination of cold working and heat treatments. As can be seen from Table 1, the Mintek alloy has a hardness of HV 170 in the cold worked condition and HV 274 after ageing. Such ageing heat treatments are typically carried out in the 300-400C range. The as-cast items, which have a hardness value of HV 87, can subsequently be hardened to in excess of HV 230 by heat treatments alone. These values exceed the performances of conventional 22–karat golds, the hardnesses of which cannot be greatly improved by any heat treatments.
Casting Properties
Casting is the most popular process for jewelry production. Various pieces of equipment and techniques, like investment casting in air or vacuum, can be used for the hard 22-karat alloy. Conventional centrifugal investment casting has been successfully conducted by melting the alloy in air with a torch and using a protective flux, such as Veri Flux from Degussa.
Color
In developing this new alloy, great care was taken so as not to change the rich yellow color of the conventional 22-karat gold. Various compositions were investigated in order to obtain a good match between color and hardness. The color of the investigated alloys was measured with a Pacific Scientific spectrophotometer and a standard CIE Lab system. Figure 2 shows the color of Mintek’s alloy in relation to that of different conventional alloys.
The CIE Lab method expresses the color as three-dimensional coordinates: L*, a*, and b*, where L* is the luminance (brightness). An L* value of 0 means that no light is reflected by the sample and an L* value of 100 means that all incident light is reflected. The a* coordinate measures the intensity of the green (negative) or red (positive) component of the spectrum, while b* measures the intensity of the blue (negative) or yellow (positive) component. The color of a sample can be defined by plotting these coordinates as a point in a three-dimensional space. Figure 2 only displays the a* and b* co-ordinates, however, as the L* coordinate was similar to that of the conventional 22- karat alloy. The color coordinates of Mintek’s alloy are very close to those of the conventional 22-karat alloy.
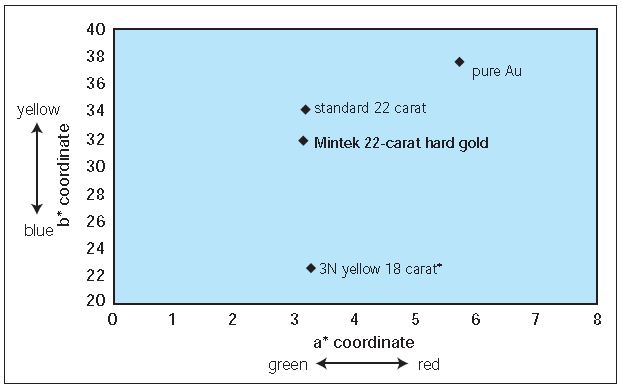
Figure 2 – Comparison of color of Mintek’s hard 22-karat gold alloy with conventional gold alloys
Workability
Trials performed on the new hard 22- karat alloy showed that a high hardness value, HV 233, could be achieved by heat treatments only.
However, jewelry manufacturing usually involves a certain degree of deformation, which also increases the hardness of the material. Mintek’s hard 22-karat alloy responds very well to deformation, as revealed by the cold working curve in Figure 3.
Figure 3 shows that, unlike the conventional alloy, Mintek’s hard 22- karat alloy undergoes an appreciable increase in hardness after as little as 25% reduction, HV 140 compared to HV 100 for the conventional 22-karat. Values in excess of HV 180 can be achieved by cold rolling the sample to 90% reduction. Heat treatments applied after 90% cold rolling can increase the hardness by almost another HV 100. The alloy could easily be reduced to more that 99,99% without intermediate annealing and cracking.
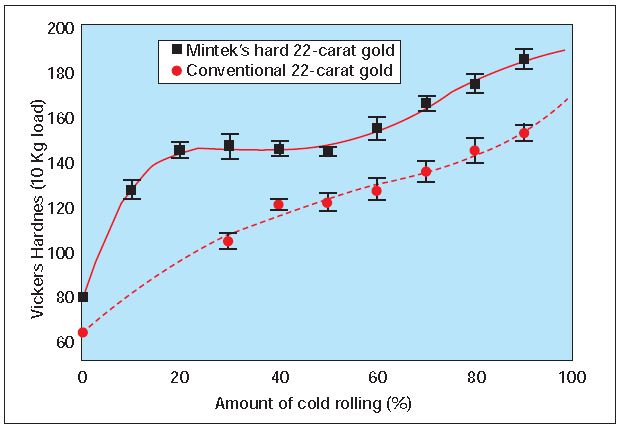
Figure 3 – Cold working curves for conventional 22-karat and Mintek’s hard 22-karat gold alloy (95% confidence interval)
Tarnish Resistance
Pure gold shows good corrosion and tarnish resistance and it preserves its rich yellow color very well. Gold alloys will also have this characteristic and, especially, high karat alloys like 22-karat gold can be expected to keep their yellow color without fear of discoloration.
Tarnish is also produced by exposure to high temperatures as is the case when heat treating samples in air. The alloying elements react with the oxygen in the air at elevated temperatures to form an oxide layer on the surface of the alloy. In order to quantify the amount of tarnish produced during heat treating, samples of Mintek’s hard 22–karat gold alloy were heat treated in air at a constant temperature for different intervals of time. A conventional 22– karat sample was used for comparison. Each sample was polished to a 0,25 m finish and the color was measured before the heat treatment.
The color of the samples was measured at different intervals during the heat treatment (without polishing) using the Pacific Scientific spectrophotometer and the standard CIE Lab system. The change in color can be quantified using a color index, DE, which can be expressed as (8):
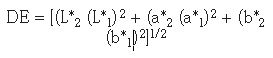
The subscript 1 refers to the color of the original polished sample and the subscript 2 to the color measurements at different times during the heat treatment. A DE value of 1 is barely discernible by the human eye, whereas color changes of more than 10 are considered to be easily noticeable (3).
Figure 4 reveals that there is no significant difference between the oxidation tendencies of conventional 22-karat gold and the hard 22-karat gold alloy. After only 20 minutes, both samples were covered with a shaded black layer of oxide, which was however easily removed by polishing.
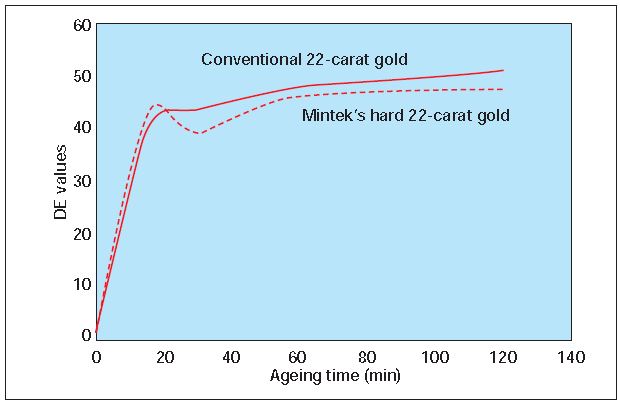
Figure 4 – Tarnish behavior of Mintek’s hard 22-karat gold alloy and conventional 22- karat gold, after heating in air.
Corrosion Resistance
Jewelry pieces are usually exposed to certain corrosive environments such as sea water and perspiration. The resistance of the hard 22-karat gold alloy to corrosion was evaluated by measuring mass loss during long term exposure to three solutions: artificial sweat (0.75 g NaCl; 0.12 g KCl; 0.27 ml lactic acid; 0.1 g urea; and H2O up to 100 ml), artificial sea water (3.56% NaCl), and dilute sulphuric acid (1 M H2SO4). The tests were carried out in duplicate, at room temperature. The samples were weighed before being immersed in the solutions and then weighed weekly for a period of two months. For comparison, conventional 22- karat gold samples were also tested.
None of the samples suffered changes in mass, and thus the presence of the alloying elements used in the Mintek 22 kt alloy does not affect the corrosion behavior of the 22-karat gold alloys in the environments evaluated.
Soldering
Soldered joints are areas where wearing and failure occur most. In order to evaluate the behavior of the Mintek alloy, pieces of the hard 22- karat alloy were soldered together using Degussa 22-karat gold solder. Because a high hardness indicates good wear resistance (9), hardness measurements were taken across the soldered joint to compare it with the rest of the alloy, Table 2. The soldered samples were heat treated according to the specifications. Figure 5 shows the hardness profile of the joint.
The measurements revealed that the hardness varies across the joint and is lowest at the center of the joint. Very good joining was obtained as all hardness values are higher than those of a conventional 22-karat alloy.
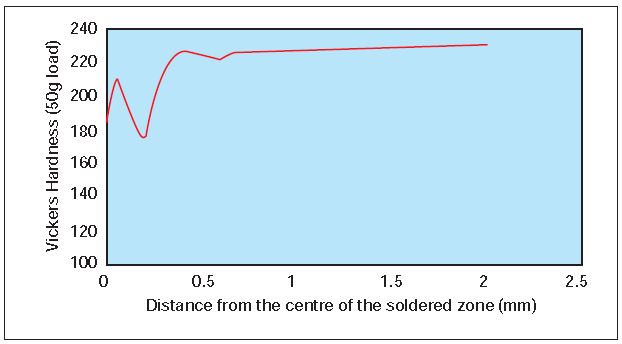
Figure 5 – Hardness profile of a soldered joint of hard 22-karat gold
Recycling
The chemical composition of the alloy, after six consecutive meltings in an arc furnace, was analyzed by Inductively Coupled Plasma/Optical Emission Spectroscopy (ICP). The results revealed negligible changes in the alloy composition. Common practice and equipment can be used for refining the Mintek alloy.
Conclusions
Casting
The new hard alloy can be cast in intricate shapes, both in air and vacuum, with optimal results. Further heat treatment can increase its hardness to more than HV 230. Deformation: The new hard alloy can be rolled down to 99,99% of its thickness without cracking. An important increase in hardness can be achieved after only 25% deformation.
Hardness
Hardness values comparable to those of conventional 18-karat gold alloys (in excess of HV 270 can be achieved by a combination of heat treatment and deformation.
Color
The Mintek hard 22-karat gold has a very good color, similar to that of conventional 22-karat gold. Tarnish resistance: Tests performed on both conventional and hard 22- karat alloys revealed similar tarnish behavior. The oxide layer formed was easily removed and it did not affect the final properties of the alloy.
Corrosion Resistance
The corrosion resistance of the alloy is similar to that of conventional 22-karat gold. Soldering: The alloy can be soldered using existing solders. The soldered joint revealed a very good hardness, which was higher than that of the conventional 22-karat gold.
Equipment
The alloy developed is user friendly, and standard equipment and practice can be used. This makes the alloy attractive to jewelry producers.
Health Hazards
No elements that may be hazardous to the human health are used in manufacturing the Mintek 22-karat alloy.
Acknowledgements
This paper is published by permission of Mintek. Our sincere thanks to Mr Pierre Ellis for his invaluable guidance and help.
References
- ‘Gold 1997’, Gold Fields Mineral Services Ltd, May 1998, p 37
- ‘Workshop Notes’, Gold Technology, No. 24, September 1998, p 26.
- M. du Toit, “The Development Of A 24 Carat Gold Alloy With Increased Hardness”, Proc. Santa Fe Symposium on Jewelry Manufakturing Technology 1997, pub. Met Chem Research Inc, 1997, p.38
- A. Nishio, “The Development Of High Strength Pure Gold”, Gold Technology, No. 19, July 1996, p 11
- C.W. Corti, “Metallurgy of Microalloyed 24 Carat Golds”, Proc. Santa Fe Symposium, 1999, p 379, publ. Met-Chem Research Inc, 1999; Gold Bulletin, 32 (2), 39 – 47, 1999.
- Gold Alloy Data Sheet for standard yellow 22 kt gold, Gold Technology, No 1, July 1990, p 6
- G. Raykhtsaum, and D.P. Agarwal, “The Color of Gold”, Gold Technology 22, July 1997, 26.
- G. Raykhtsaum, and D.P. Agarwal, “Measuring Tarnish. Tarnish Behaviour of Low Karat Jewelry Alloys – Quantitative Analysis”, American Jewelry Manufakturers, February 1990, 116-119, 147
- T. Biggs, S. Taylor, K.L. Rutherford, and I.M. Hutchings, “The Wear Behavior of Some Platinum Alloys”, Precious Metals 1997, IPMI Conference, San Francisco, 331
Source: World Gold Council